Being the go-to science guy, I get questions like: so can something be done about my hair loss?
The context of this is that I frequently talk about replication crisis, and pharma companies cheating with studies, most things sold in pharmacies not working etc., so the prior is skeptical. Still, some treatments do work. Are the ones for male hair loss among them? I think so!
As explained before, we first try the Cochrane database. Actually, first we google “hair loss” and learn that the medical term for this issue is androgenetic alopecia, or male-related/pattern hair loss/baldness. Finding the right medical paper often depends on searching for the right medical term. And then we search:
But there are no Cochrane reviews (top left tab), so this is no use. There’s a lot of trials found, so meta-analyses should exist. So we try Google Scholar, and look for newest meta-analyses/systematic reviews: “androgenetic alopecia” meta analysis. (We use the quotes here to avoid other conditions also called something with androgenic or alopecia.) First meta:
- Adil, A., & Godwin, M. (2017). The effectiveness of treatments for androgenetic alopecia: a systematic review and meta-analysis. Journal of the American Academy of Dermatology, 77(1), 136-141.
Their forest plots tell us more than the abstract:
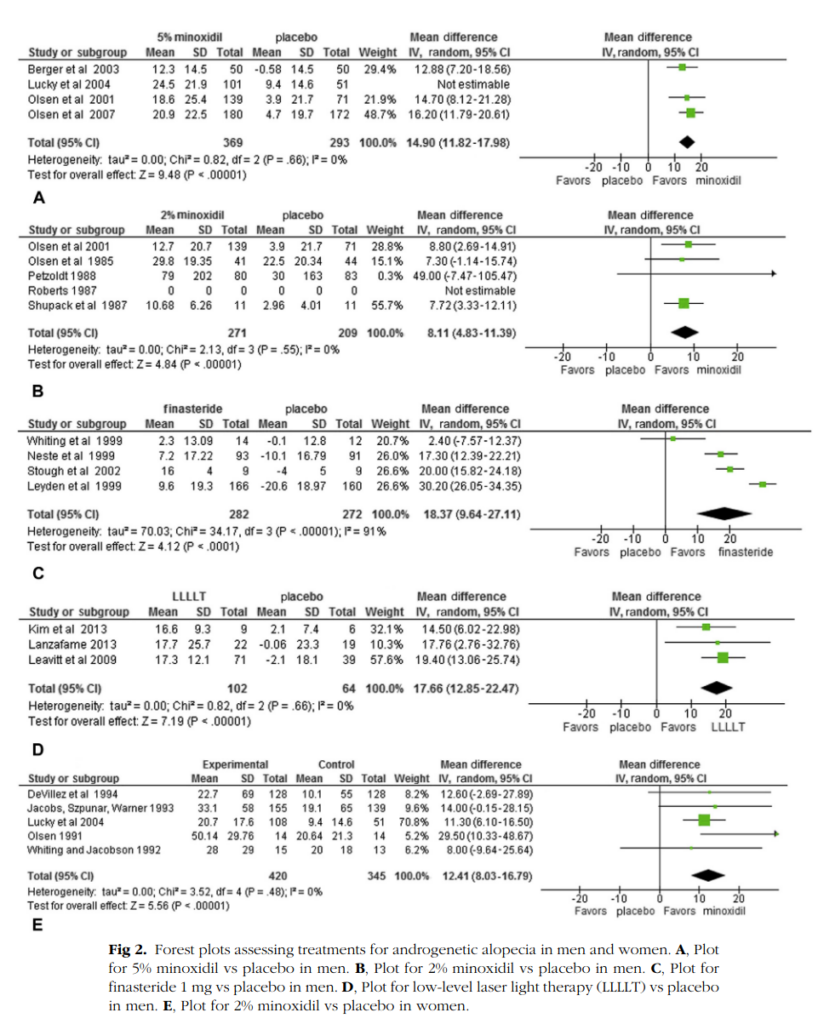
The different parts are different treatments vs. placebo (i.e. no/fake treatment). We are struck by the poverty of studies. The most used medical ailments are minoxidil (brand name Rogaine) and finasteride (brand names Proscar, Propecia). Yet despite widespread use, there’s only 4, 5, and 4 studies for these! (9 total studies for minoxidil at doses of 2% or 5%). The results do not immediately smell of p-hacking. The largest studies are Olsen et al 2007 and Leyden et al 1999. Let’s look at those:
- Olsen, E. A., Whiting, D., Bergfeld, W., Miller, J., Hordinsky, M., Wanser, R., … & Kohut, B. (2007). A multicenter, randomized, placebo-controlled, double-blind clinical trial of a novel formulation of 5% minoxidil topical foam versus placebo in the treatment of androgenetic alopecia in men. Journal of the American Academy of Dermatology, 57(5), 767-774.
Background An alternative to currently marketed topical minoxidil solutions is desirable.
Objective To assess the efficacy and safety of a new 5% minoxidil topical formulation in a propylene glycol–free foam vehicle in men with androgenetic alopecia (AGA).
Methods This was a 16-week, double-blind, placebo-controlled trial of 5% minoxidil topical foam (MTF) in 352 men, 18 to 49 years old. At week 16, 143 subjects continued on an open-label phase to collect 52 weeks of safety information on 5% MTF.
Results At week 16 compared with baseline, there was a statistically significant increase in (1) hair counts in the 5% MTF group versus placebo (P < .0001) and (2) subjective assessment of improved hair loss condition (P < .0001) in the 5% MTF group versus placebo. The 5% MTF was well tolerated over a 52-week period.
Limitations There was no collection of efficacy data beyond 16 weeks.
Conclusions We believe that 5% MTF is a safe and effective treatment for men with AGA.Supported by Pfizer Inc. Disclosure: Drs Olsen, Whiting, Bergfeld, Miller, and Hordinsky received study grants; Drs Olsen and Miller also received other grant support. Drs Olsen, Whiting, Bergfeld, Miller, and Hordinsky have served as consultants. Ms Wanser and Drs Zhang and Kohut were all employees of Pfizer, Inc at the time the study was conducted and during the preparation of the manuscript.
- Leyden, J., Dunlap, F., Miller, B., Winters, P., Lebwohl, M., Hecker, D., … & Waldstreicher, J. (1999). Finasteride in the treatment of men with frontal male pattern hair loss. Journal of the American Academy of Dermatology, 40(6), 930-937.
Background: Finasteride, a specific inhibitor of type II 5α-reductase, decreases serum and scalp dihydrotestosterone and has been shown to be effective in men with vertex male pattern hair loss. Objective: This study evaluated the efficacy of finasteride 1 mg/day in men with frontal (anterior/mid) scalp hair thinning. Methods: This was a 1-year, double-blind, placebo-controlled study followed by a 1-year open extension. Efficacy was assessed by hair counts (1 cm2 circular area), patient and investigator assessments, and global photographic review. Results: There was a significant increase in hair count in the frontal scalp of finasteride-treated patients (P < .001), as well as significant improvements in patient, investigator, and global photographic assessments. Efficacy was maintained or improved throughout the second year of the study. Finasteride was generally well tolerated. Conclusion: In men with hair loss in the anterior/mid area of the scalp, finasteride 1 mg/day slowed hair loss and increased hair growth.
The reported clinical study was entirely supported by Merck Research Laboratories, Merck & Co, Inc. Merck & Co, Inc is the company that manufactures and markets finasteride.
The p values are good, and they are done in western countries. Both are industry sponsored studies, which makes us more cautious. But since industry cheats the same way normal researchers do (p-hacking), and these studies are not p hacked (both main results are p < .001), then this fact does not seem to affect our belief much. The large studies are in line with the smaller studies, so there doesn’t seem any obvious publication bias, but with 4-5 studies, we can’t really say so with any confidence.
Any later, comprehensiver meta-analyses? Well there’s this one that focuses on the combined treatment of both drugs above:
- Chen, L., Zhang, J., Wang, L., Wang, H., & Chen, B. (2020). The efficacy and safety of finasteride combined with topical minoxidil for androgenetic alopecia: a systematic review and meta-analysis. Aesthetic plastic surgery, 44(3), 962-970.
Background The combination of finasteride and topical minoxidil has been used for treating patients with androgenetic alopecia (AGA). However, whether combining these two medications results in greater efficacy than monotherapy is a question worth exploring.
Objective This meta-analysis aims to determine the efficacy and safety of combined treatment of finasteride and topical minoxidil.
Methods A comprehensive search of the Embase, PubMed, and the Cochrane Library databases was performed. Data were extracted and analyzed according to predefined clinical endpoints.
Results Five randomized controlled trials (RCTs) were included in our meta-analysis. All studies compared combined therapy with minoxidil, but only 2 RCTs compared combined therapy with finasteride. Compared with minoxidil or finasteride alone, the combined group had a significantly higher global photographic evaluation score (P < 0.00001), more patients with marked improvement (P < 0.001), and fewer patients with deterioration or no change (P < 0.001). There was no significant difference between the combined group and minoxidil- or finasteride-only groups in the number of patients with moderate and mild improvements, hair density change, or adverse events.
Conclusions In patients with AGA, the combination treatment of finasteride and topical minoxidil has better therapeutic efficacy than and similar safety as monotherapy. However, the best concentration of combination treatment requires further studies with sound methodological quality.
Only 5 studies in this meta-analysis!
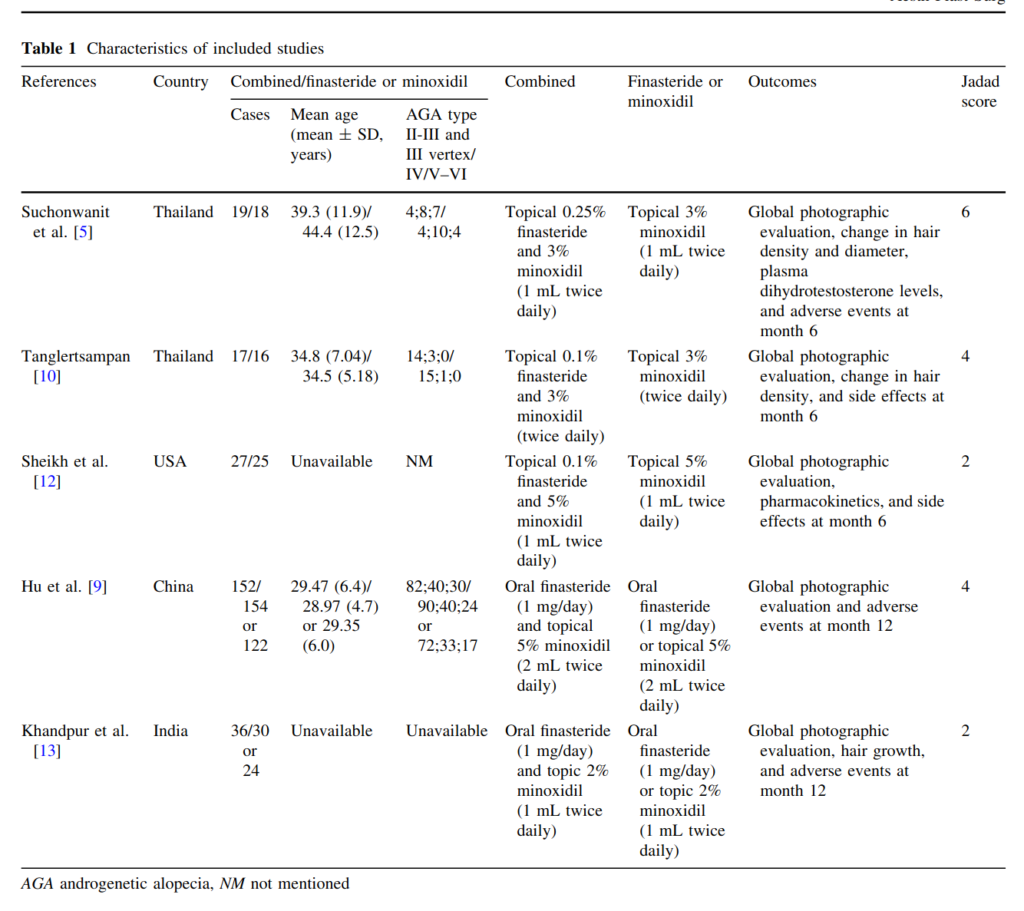
Of the 5 studies, 4 are in dubious countries. The US study is too small to care much about (52 persons). This leaves us with the Chinese study Hu et al 2015:
Finasteride at 1 mg/day and 5% topical minoxidil are effective in male androgenetic alopecia (MAGA). However, studies describing their effects in Chinese individuals are scarce. 450 Chinese MAGA patients were randomly assigned to receive finasteride (n = 160), minoxidil (n = 130) and combined medication (n = 160) for 12 months. The patients returned to the clinic every 3 months for efficacy evaluation. And efficacy was evaluated in 428 men at treatment end, including 154, 122, and 152 in the finasteride, 5% minoxidil, and combination groups, respectively. All groups showed similar baseline characteristics, including age at enrollment, and duration and severity of alopecia (p > 0.05). At 12 months, 80.5, 59, and 94.1% men treated with finasteride, 5% minoxidil and the combination therapy showed improvement, respectively. Adverse reactions were rare (finasteride, 1.8%; minoxidil, 6.1%), and disappeared right after drug withdrawal. In conclusion, finasteride is superior to 5% minoxidil, while the combined medication showed the best efficacy.
MAGA! I mean…
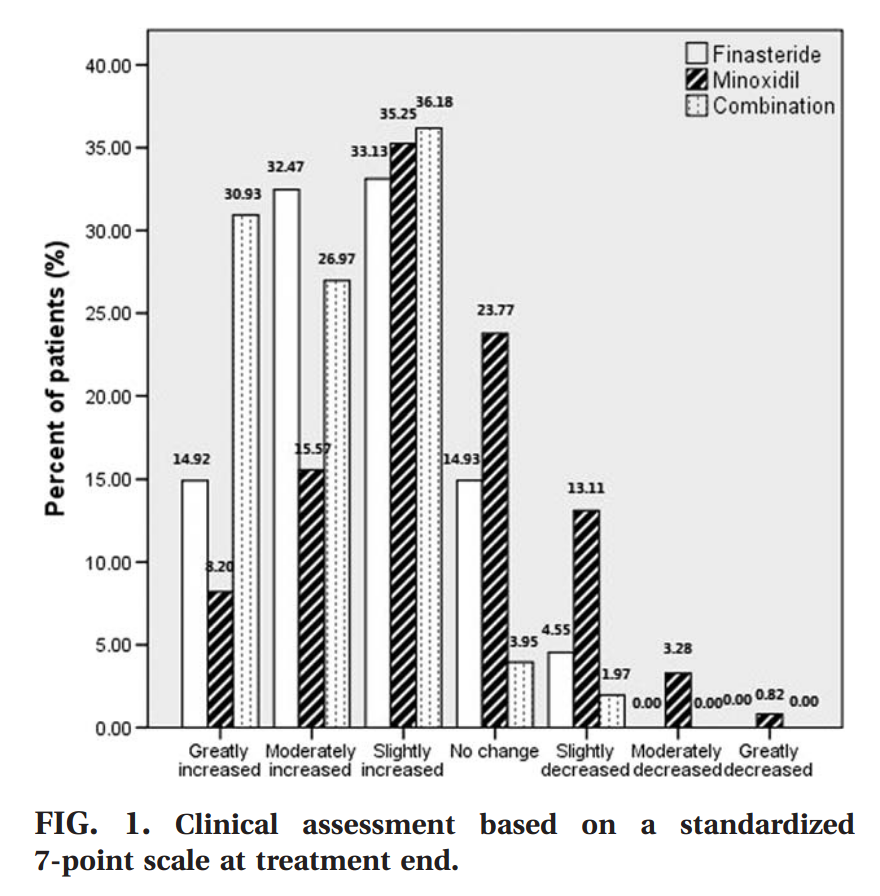
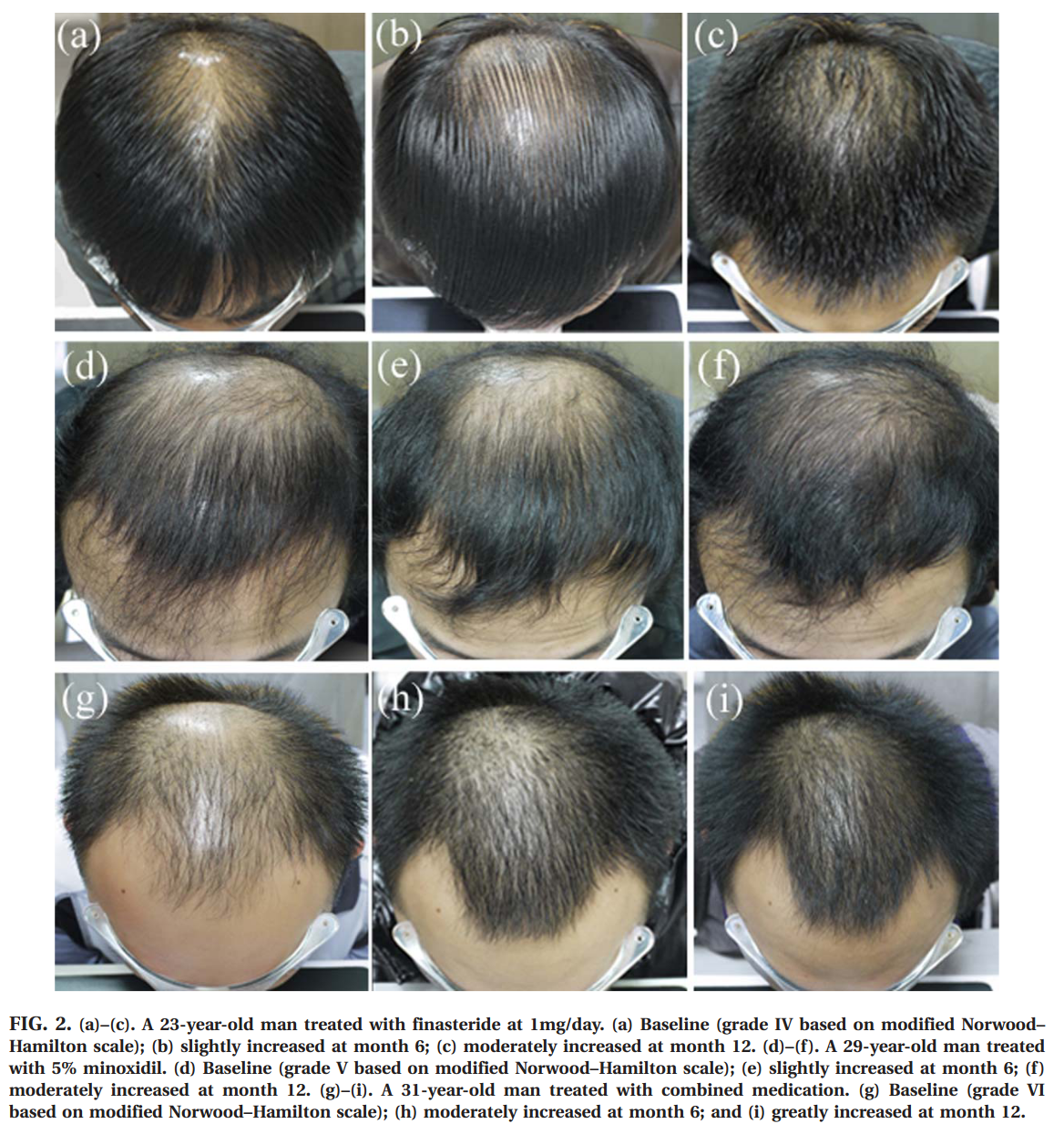
Hair growth rate in the finasteride group at month 3 was similar to that of minoxidil treated individuals (finasteride: 39.0% vs. minoxidil: 38.5%, p > 0.05), and reduced compared with the combination group (54.6%, p < 0.01). At months 6, 9, and 12, the patients taking finasteride showed a more pronounced improvement com- pared with those applying minoxidil (p < 0.01); the efficacy of the combined treatment remained the best (p < 0.01). The rating scores of global photographic evaluation are shown in FIG. 1. FIG. 2 exhibits the changes in hair growth from base- line by means of global photographic assessment.
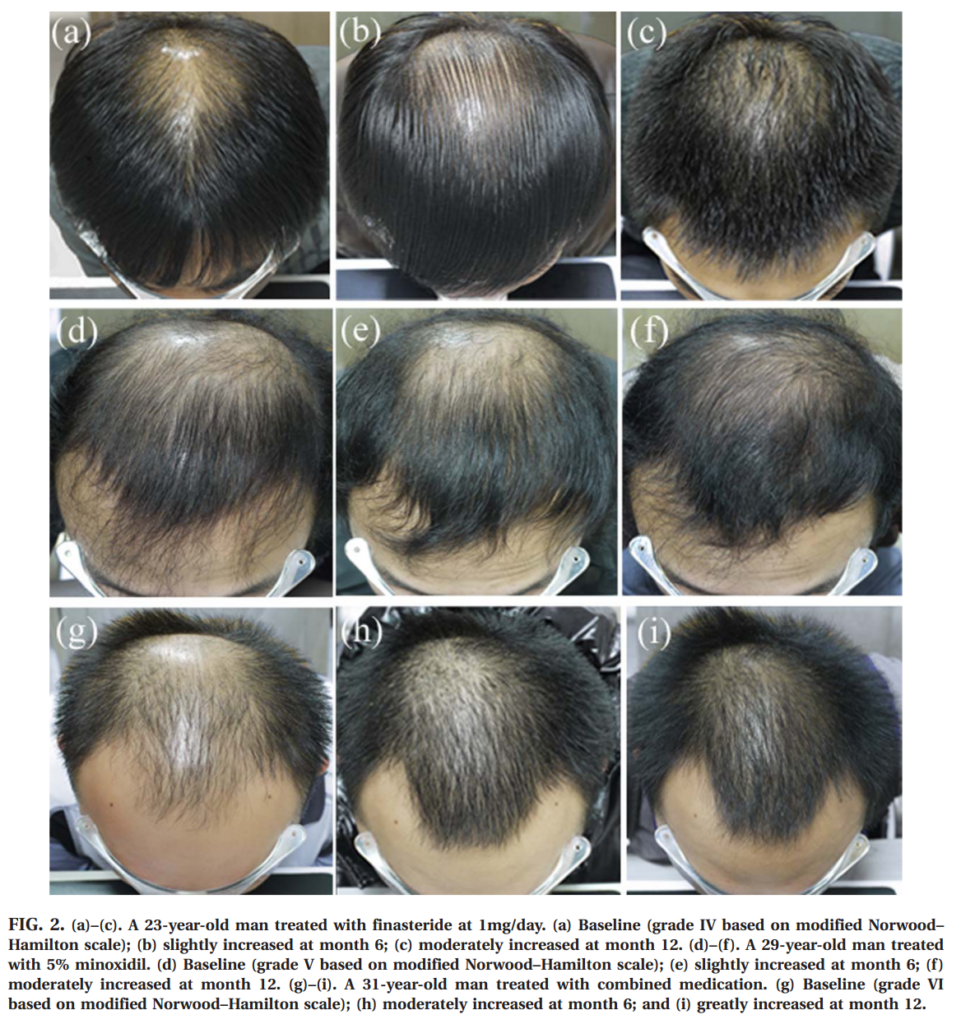
They also have scalp photos, and we love scalps on this blog:
There’s also this more impressive sounding meta-analysis:
- Gupta, A. K., Mays, R. R., Dotzert, M. S., Versteeg, S. G., Shear, N. H., & Piguet, V. (2018). Efficacy of non‐surgical treatments for androgenetic alopecia: a systematic review and network meta‐analysis. Journal of the European Academy of Dermatology and Venereology, 32(12), 2112-2125.
Androgenetic alopecia, or male/female pattern baldness, is the most common type of progressive hair loss disorder. The aim of this study was to review recent advances in non-surgical treatments for androgenetic alopecia and identify the most effective treatments. A network meta-analysis (NMA) was conducted of the available literature of the six most common non-surgical treatment options for treating androgenetic alopecia in both men and women; dutasteride 0.5 mg, finasteride 1 mg, low-level laser therapy (LLLT), minoxidil 2%, minoxidil 5% and platelet-rich plasma (PRP). Seventy-eight studies met the inclusion criteria, and 22 studies had the data necessary for a network meta-analysis. Relative effects show LLLT as the superior treatment. Relative effects show PRP, finasteride 1 mg (male), finasteride 1 mg (female), minoxidil 5%, minoxidil 2% and dutasteride (male) are approximately equivalent in mean change hair count following treatment. Minoxidil 5% and minoxidil 2% reported the most drug-related adverse events (n = 45 and n = 23, respectively). The quality of evidence of minoxidil 2% vs. minoxidil 5% was high; minoxidil 5% vs. placebo was moderate; dutasteride (male) vs. placebo, finasteride (female) vs. placebo, minoxidil 2% vs. placebo and minoxidil 5% vs. LLLT was low; and finasteride (male) vs. placebo, LLLT vs. sham, PRP vs. placebo and finasteride vs. minoxidil 2% was very low. Results of this NMA indicate the emergence of novel, non-hormonal therapies as effective treatments for hair loss; however, the quality of evidence is generally low. High-quality randomized controlled trials and head-to-head trials are required to support these findings and aid in the development of more standardized protocols, particularly for PRP. Regardless, this analysis may aid physicians in clinical decision-making and highlight the variety of non-surgical hair restoration options for patients.
Why impressive? Well, instead of only doing two-way comparisons, it includes all the pairwise comparisons. Some of these can provide indirect information about each other. Consider the simple situation: you have drug A vs. placebo, and drug B vs. placebo. Since the placebo effect should be identical across studies, you can compare drug A’s relative benefit vs. to placebo to drug B’s to placebo as a way to compare drug A and B without having a study with drug A and B together. This logic can be extended to any set of pairwise comparisons, including ones never observed. The authors did this, and produced this table:
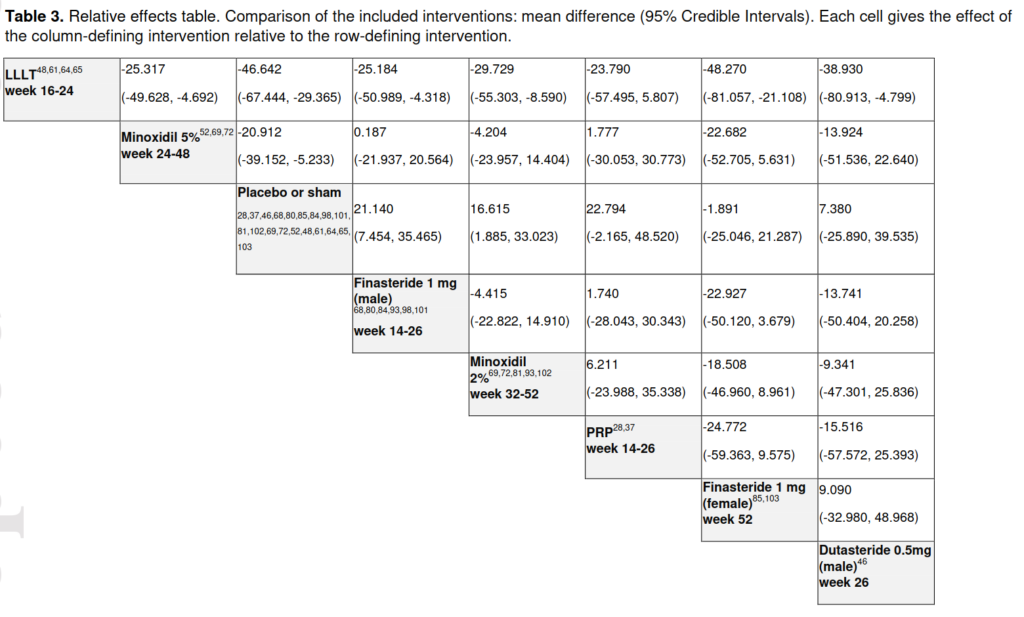
According to their findings, the laser treatment does overall best (LLLT = low-level laser therapy; PRP = platelet-rich plasma). (Dutasteride is just a similar drug to finasteride, which are both blockers of of a testosterone derivative (5α-Reductase inhibitor). I would not really consider it alone, also because there’s only 1 study of it included here.) One can even have a Bayesian rank ordering of treatments:
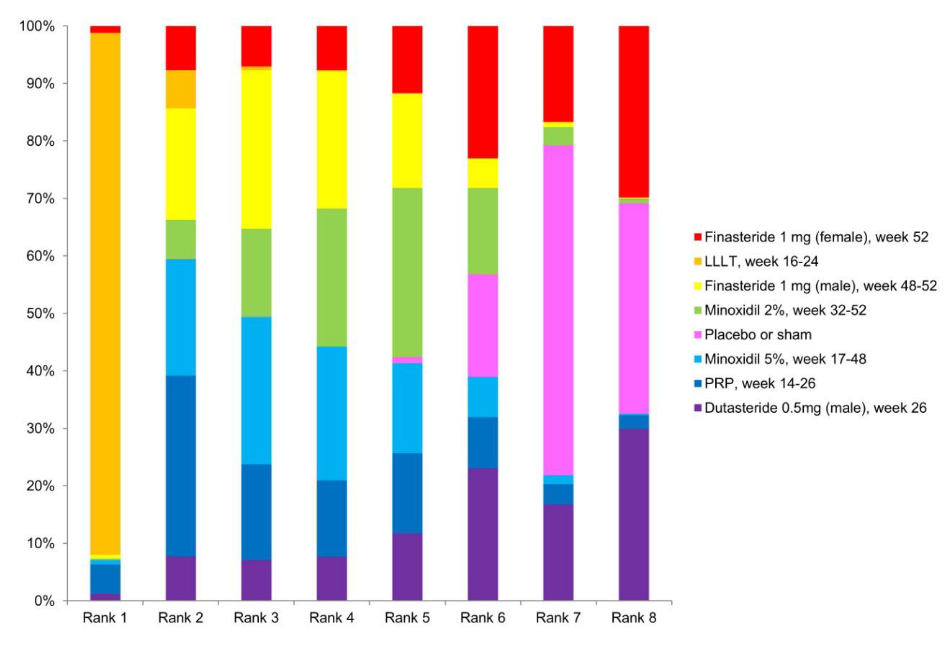
The LLLT treatment was also mentioned earlier, but I ignored it because the sample sizes of the studies were too small. The studies they included here are not any more convincing:
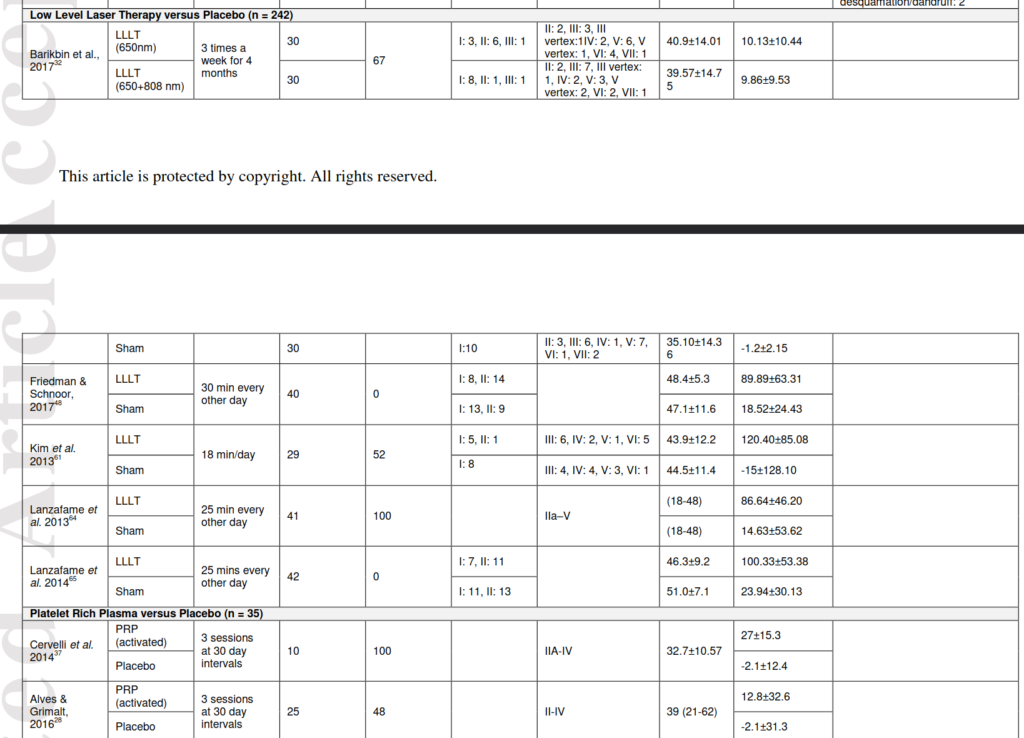
The reason I ignore small studies is that findings of various replicability projects find that smaller studies are more inflated. This is a consequence of p-hacking, that smaller studies with p < .05 produce more inflated estimates. The sample sizes for the LLLT studies are at most 42 persons, and for PRP, at most 25. In Bayesian terms, I think they should use a study-level shrinkage factor (skeptical prior) that reduces effect sizes towards 0 based on sample size/precision. That would reduce this bias.
There are more meta–analyses, but they are generally just combining the same studies in different ways, so reading them is of limited value if we are already skeptical of doing many comparisons of small datasets.
Conclusions
I am not a doctor, don’t sue me.
Based on my reading, I suggest trying finasteride and minoxidil. Finasteride has been on market since 1992, so it is well tested, and you can be pretty sure no surprise very bad side effects. I have some friends who also studied some of this evidence, tried them out, and liked the results, so my anecdata fit the data.