I know some people are who are into conspiratorial thinking about COVID vaccines. Bill Gates trying to decrease your fertility stuff. Anyway, so I did the science thing, that is, I used a neutral search on an academic search engine to locate actual data on the question. Specifically, Google Scholar with the query “Covid vaccine fertility” limited to 2021 studies. I tweeted what I found. I also looked at the studies citing any of the included studies. This post is the more permanent blogpost version.
-
Male, V. (2021). Are COVID-19 vaccines safe in pregnancy?. Nature Reviews Immunology, 1-2.
As the COVID-19 vaccination programme starts to be rolled out, many young women are hesitant to accept the vaccine, citing concerns about fertility. Meanwhile, those offered the vaccine during pregnancy must decide whether they will accept, even though pregnant people were excluded from the clinical trials. Data on accidental pregnancies that occurred during the trials and, increasingly, outcomes in pregnant people who receive the vaccine can help these groups to make informed decisions.
Specifically, they have some data:
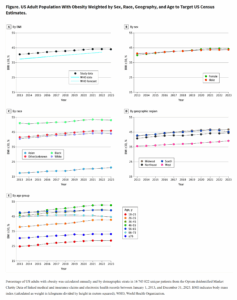
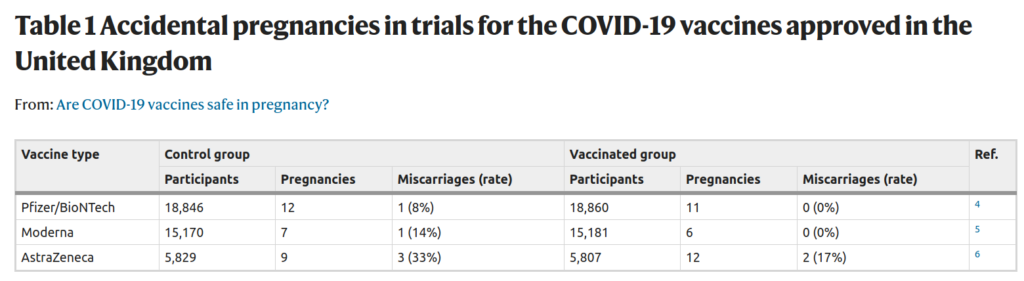
But the data that speak most clearly to the question of whether the COVID-19 vaccines harm fertility come from the clinical trials themselves4,5,6. Developmental and reproductive toxicity studies show that the vaccines do not prevent female rodents becoming pregnant or harm the pups if given during pregnancy. We also have an idea of how the vaccines affect pregnancy in humans from the volunteers who became pregnant during the clinical trials. Pregnant people were excluded from the trials and participants were asked to avoid becoming pregnant, but, nonetheless, 57 pregnancies occurred across the trials of the three vaccines that have so far been approved in the UK. The outcomes of these pregnancies, so far as they have progressed, are summarized in Table 1. There was no significant difference in the rate of accidental pregnancies in the vaccinated groups compared with the control groups, which indicates that the vaccines do not prevent pregnancy in humans. Similarly, the miscarriage rates are comparable between the groups, indicating no detrimental effect of vaccination on early pregnancy.
One can do the formal stats here if desired, but the non-vaccinated group had somewhat higher rate of miscarriage, and the pregnancy rates were also about the same. From another angle, we can also look at people using IVFs. There are at least 2 studies that use before and after design here comparing the fertility parameters of the same people. These studies are small, so they can’t rule out any medium or small effects, but they do make it very unlikely there are major negative effects.
-
Safrai, M., Reubinoff, B., & Ben-Meir, A. (2021). BNT162b2 mRNA Covid-19 vaccine does not impair sperm parameters. medRxiv.
Mass vaccination using newly approved vaccines against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has begun globally. However, their effect on fertility have not yet been investigated. Previous studies demonstrate that SARS-CoV-2 infection may impair sperm parameters. In this study, we are the first to assess the effect of the BNT162b2 mRNA Covid-19 vaccine on sperm parameters. Our results demonstrate that the vaccine does not impair sperm parameters. Thus, we recommend that couples desiring to conceive should vaccinate, as vaccination does not affect sperm whereas SARS-CoV-2 infection does impair sperm.
n=43
- Orvieto, R., Noach-Hirsh, M., Segev-Zahav, A., Haas, J., Nahum, R., & Aizer, A. (2021). Does mRNA SARS-CoV-2 vaccine influence patients’ performance during IVF-ET cycle?. Reproductive Biology and Endocrinology, 19(1), 1-4.
Objective No information exists in the literature regarding the effect of mRNA SARS-CoV-2 vaccine on subsequent IVF cycle attempt. We therefore aim to assess the influence of mRNA SARS-CoV-2 vaccine on IVF treatments.
Design An observational study.
Setting A tertiary, university-affiliated medical center.
Patients and Methods All couples undergoing consecutive ovarian stimulation cycles for IVF before and after receiving mRNA SARS-CoV-2 vaccine, and reached the ovum pick-up (OPU) stage. The stimulation characteristics and embryological variables of couples undergoing IVF treatments after receiving mRNA SARS-CoV-2 vaccine were assessed and compared to their IVF cycles prior to vaccination.
Main outcome measures Stimulation characteristics and embryological variables.
Results Thirty-six couples resumed IVF treatment 7–85 days after receiving mRNA SARS-CoV-2 vaccine. No in-between cycles differences were observed in ovarian stimulation and embryological variables before and after receiving mRNA SARS-CoV-2 vaccination.
Conclusions mRNA SARS-CoV-2 vaccine did not affect patients’ performance or ovarian reserve in their immediate subsequent IVF cycle. Future larger studies with longer follow-up will be needed to validate our observations.
n=36
-
Bentov, Y., Beharier, O., Moav-Zafrir, A., Kabessa, M., Godin, M., Greenfield, C., … & Klement, A. H. (2021). Ovarian follicular function is not altered by SARS-Cov-2 infection or BNT162b2 mRNA Covid-19 vaccination. medRxiv.
Objective To characterize anti-COVID-19 antibodies in follicular fluid and compare ovarian follicle function in women following confirmed SARS-CoV-2 infection, COVID-19 vaccination, and non-infected, unvaccinated controls.
Design This is a cohort study conducted between February 1 and March 10, 2021.
Setting A single university hospital-based IVF clinic.
Participants Consecutive sample of female patients undergoing oocyte retrieval.
Interventions Consenting patients were recruited and assigned to one of three study groups: recovering from confirmed COVID 19 (n=9); vaccinated (n=9); and uninfected, non-vaccinated controls (n=14). Serum and follicular fluid samples were taken and analyzed for anti-COVID IgG as well as estrogen, progesterone and HSPG2 concentration, as well as the number and maturity of aspirated oocytes and previous estrogen and progesterone measurements.
Main outcome measures Follicular function, including steroidogenesis, follicular response to the LH/hCG trigger, and oocyte quality biomarkers.
Results Both natural and vaccine elicited anti-COVID IgG antibodies were detected in the follicular fluid in levels proportional to the IgG serum concentration. No differences were detected in any of the surrogate ovarian follicle quality reporting parameters.
Conclusions and relevance Both SARS-COV-2 infection and vaccination with the BNT162b2 mRNA vaccine mediate IgG immunity that crosses into the follicular fluid. No detrimental effect on follicular function was detected.
n=32
Many people claim some kind of cross-reactivity to syncytin-1, but this apparently can’t be found.
-
Mattar, C. N., Koh, W., Seow, Y., Hoon, S., Venkatesh, A., Dashraath, P., … & Tambyah, P. A. (2021). Addressing anti-syncytin antibody levels, and fertility and breastfeeding concerns, following BNT162B2 COVID-19 mRNA vaccination. medRxiv.
Objective To determine whether antibodies against the SARS-CoV-2 spike protein following BNT162B2 (Pfizer-BioNTech) COVID-19 mRNA vaccination cross-react with human syncytin-1 protein, and if BNT162B2 mRNA enters breast milk.
Methods In this observational cohort study of female front-line workers with no history of COVID-19 infection, we amplified BNT162B2 mRNA in plasma and breast milk and assayed anti-SARS-CoV-2 neutralising antibodies and anti-human syncytin-1 binding antibodies in plasma, at early (1-4 days) and late (4-7 weeks) time points following first-dose vaccination.
Results Fifteen consented participants (mean age 40.4 years, various ethnicities) who received at least one dose of BNT162B2, including five breast-feeding women and two women who were inadvertently vaccinated in early pregnancy, were recruited. BNT162B2 mRNA, detected by amplifying part of the spike-encoding region, was detected in plasma 1-4 days following the first dose (n=13), but not 4-5 weeks later (n=2), nor was the mRNA isolated from aqueous or lipid breast milk fractions collected 0-7 days post-vaccination (n=5). Vaccine recipients demonstrated strong SARS-CoV-2 neutralising activity by at least four weeks after the first dose (n=15), including the two pregnant women. None had placental anti-syncytin-1 binding antibodies at either time-point following vaccination.
Conclusions BNT162B2-vaccinated women did not transmit vaccine mRNA to breast milk, and did not produce a concurrent humoral response to syncytin-1, suggesting that cross-reactivity to syncytin-1 on the developing trophoblast, or other adverse effects in the breast-fed infant from vaccine mRNA ingestion, are unlikely.
Jonatan Pallesen incidentally has an opposite view thread. Not with fertility outcomes, but with tissue activity and things like that. In general, I prefer fertility outcomes, a harder outcome.